| | Signs of nuisance bacteria | Shock chlorination method | Worksheet
.
| Well maintenance is essential to ensure that a well will last. |
Shock chlorination is a relatively inexpensive and straightforward procedure used to control bacteria in water wells. Many types of bacteria can contaminate wells, but the most common are iron-related (IRB) and sulfate-reducing bacteria (SRB). Although not a cause of health problems in humans, these nuisance bacteria may coat the inside of the well casing, water piping and pumping equipment, creating problems such as:
- Reduced well yield
- Restricted water flow in distribution lines
- Staining of plumbing fixtures and laundry
- Plugging of water treatment equipment
- "Rotten egg” odour.
- Increased corrosion of the metal parts of the well and distribution system
Wells can also be contaminated with harmful bacteria such as fecal coliforms or E. coli. Shock chlorination is the most effective method to eliminate them. Bacteria may be introduced during drilling of a well or when pumps are removed for repair and laid on the ground. However, IRBs and SRBs (as well as other bacteria) can exist naturally in groundwater.
A well creates a direct path for oxygen to travel into the ground where it would not normally exist accelerating the growth of bacteria that require oxygen. When a well is pumped, the water flowing in will also bring in nutrients that enhance bacterial growth.
| Water wells provide ideal conditions for IRBs. To thrive, IRBs require 0.5-4 mg/L of dissolved oxygen, as little as 0.01 mg/L dissolved iron and a temperature range of 5 to 15�C. Some IRBs use dissolved iron in the water as a food source. |
Note: All iron staining problems are not necessarily caused by iron-related bacteria. The iron naturally present in the water can be the cause. See Module 12 "Other Resources" for more information.
Signs of Nuisance Bacteria
There are a number of signs that indicate the presence of iron-related, sulfate-reducing and other slime-producing bacteria. They include:
- Slime growth
- Rotten egg odour
- Increased staining.
Slime Growth
The easiest way to check a well and water system for nuisance bacteria is to examine the inside surface of the toilet flush tank. If you feel a greasy slime or growth, these are probably present.
Rotten Egg Odour
Sulfate-reducing bacteria can cause a rotten egg odour in water. Iron-related bacteria aggravate the problem by creating an environment that encourages the growth of SRBs in the well. SRBs prefer to live underneath the slime layer that the IRBs form because they thrive in low oxygen environments. SRBs produce hydrogen sulfide gas as a by-product, resulting in a “rotten egg” or sulfur odour in the water and may produce small amounts of acid that can corrode the well casing and pumping equipment.
Increased Staining Problems
Iron-related bacteria can concentrate iron in water sources with low iron content. It can create a staining problem where one never existed before or make an iron staining problem worse as time goes by.
Use the following checklist to determine if you have an iron-related or sulfate-reducing bacteria problem. The first three are very specific problems related to these bacteria. The last two problems can be signs of other problems as well.
 Checklist to Determine an Iron or Sulfate-Reducing Bacteria Problem Checklist to Determine an Iron or Sulfate-Reducing Bacteria Problem
- Greasy slime on inside surface of toilet flush tank
- Increased red staining of plumbing fixtures and laundry
- Sulfur odour
- Reduced well yield
- Restricted water flow
|
Shock Chlorination Method
Shock chlorination is used to control iron-related and sulfate-reducing bacteria and to eliminate faecal coliform or E. coli bacteria in a water system. To be effective, shock chlorination should be done on a regular basis at least once or twice per year as part of a routine well
maintenance program. Start the treatments early in the life of your well. Shock chlorination will disinfect the following:
- The entire well depth The formation around the bottom of the well
- The pressure system Some water treatment equipment
- The distribution system.
To accomplish this, a large volume of chlorinated water is siphoned down the well to displace all the water in the well and some of the water in the formation surrounding the well.
| Before you shock chlorinate, consult your water treatment equipment supplier to ensure the appropriate steps are taken to protect your treatment equipment. |
Effectiveness of shock chlorination
With shock chlorination, the entire system (from the water-bearing formation, through the well-bore and the distribution system) is exposed to water which has a concentration of chlorine strong enough to kill iron-related and sulfate reducing bacteria.
Bacteria collect in the pore spaces of the formation and on the casing or screened surface of the well. To be effective, you need to calculate the correct volumes of chlorine and water required to reach and disinfect the entire cased section of the well and adjacent water-bearing formation. The procedure described on the following pages does not completely eliminate nuisance bacteria from the water system, but it will hold it in check. To control the iron-related bacteria, you will have to repeat the treatment on a regular basis, likely each spring and fall as a regular maintenance procedure. If your well has never been shock chlorinated or has not been done for some time, it may be necessary to contact a licensed water well contractor about other products and methods for control of bacteria.
| Be aware that chlorinating old, unmaintained wells may uncover existing holes in steel casing and speed up inevitable well failure. |
Adding chlorine increases the pH of the water. An increase in pH reduces the ability of chlorine to disinfect or kill bacteria. Your licensed water well contractor may have specialized chemicals designed to reduce pH and optimize disinfection. Shock chlorination will not be effective on wells that have been seldom or never been treated. These poorly maintained wells likely require the services of an experienced licensed water well contractor who has the necessary equipment and products to effectively and safely clean and restore the well water quality and production. Sometimes these wells can be restored to near their original water quality and production capacity after the well driller uses scrubbing equipment and applies an acid treatment to remove the heavy layers of bacterial slime on the well casing prior
to disinfecting with a chlorine solution. After a thorough cleaning it may be possible to return to regular shock chlorination treatments to control bacteria buildup and its related problems.
Shock chlorination procedure for drilled wells
A modified procedure is also provided for large diameter wells. Caution: If your well is low yielding or tends to pump any silt or sand, you must be very careful using the following procedure because over-pumping may damage the well. When pumping out the chlorinated solution, monitor the water discharge for sediment.
Step 1
Store sufficient water to meet farm and family needs for 8 to 48 hours.
Step 2
Pump the recommended amount of water (see Table 1, Amount of Chlorine Required to Obtain a Chlorine Concentration of 200 PPM) into clean storage. A clean tank, used only for storing potable or chlorinated water should be used. The recommended amount of water to use is twice the volume of water present in the well casing*. To measure how much water is in the casing, subtract the non-pumping water level from the total depth of the well. See the example below.
*Large cisterns should be bypassed and treated separately. If the water must travel through a long or large diameter
pipeline, or large pressure tanks, an extra volume of chlorinated solution should be used. Consult with a licensed
water well drilling contractor or a water specialist. |
Imperial Example
The drilling record indicates the casing is 200 ft. in length and the non-pumping ("static") water level is 100 ft. The length of casing that is holding water in it is 100 ft. (200-100). If your casing is 6 in. in diameter you need to pump 2.4 gal. of water for every foot of water in the casing, into your storage container. Since you have 100 ft. of water in the casing, you will pump 2.4 gal./ft. x 100 ft. = 240 gal. of water into storage.
Using Table 1, calculate how much water you need to pump into clean storage.
Casing diameter_________ needs_________gal./ft. x __________ ft. = _________gal.
Table 1 Amount of Chlorine Required to Obtain a Chlorine Concentration of 200 PPM
Casing Diameter | Volume of Water Needed | 5 �%
Domestic
Chlorine Bleach | 12% Industrial
Sodium
Hypochlorite | '70%
High Test
Hypochlorite |
Water needed per 1 ft. (30 cm)
of water in the casing | L needed
per 1 ft. (30 cm)
of water | L need
per 1 ft. (30 cm)
of water | Dry weight 1
per 1 ft. (30 cm)
of water |
(in) | (mm) | (gal.) | (L) | (L) | (L) | (g) |
4 | (100) | 1.1 | 5.0 | .019 | .008 | 1.44 |
6 | (150) | 2.4 | 10.9 | .042 | .018 | 3.12 |
8 | (200) | 4.2 | 19.1 | .072 | .032 | 5.46 |
24 | (600)2 | extra 200 gal. | extra 1000 L | .340 | .148 | 25.40 |
36 | (900)2 | extra 200 gal. | extra 1000 L | .76 | .34 | 57.20 |
1 Domestic chlorine bleach should not have additives or perfumes.
2Since a dry chemical is being used, it should be mixed with water to form a chlorine solultion before placing it in the well.
3See modified procedure for large diameter wells on page 53.
4 To reduce the chlorine concentrations to 50 PPM, divide the above chlorine amounts by 4.
Sodium Hypochlorite (5�% & 12%, liquid form) has a shelf life. Purchase new stock and check for an expiry date. Buy only as much as you need for the procedure. 12% industrial sodium hypochlorite and 70% high test hypochlorite are available from:
- Water treatment suppliers
- Licensed water well contractors
- Swimming pool maintenance suppliers
- Dairy equipment suppliers
- Some hardware stores.
|
Step 3
Calculate the amount of chlorine that is required, as shown in Table 1. Mix the chlorine with the previously measured water to obtain a 200 ppm chlorine solution.
| Caution: Chlorine is corrosive and can even be deadly. |
Calculating Amount of Chlorine Example
If your casing is 6 in. and you are using 12% industrial sodium hypochlorite, you will require 0.018 L per ft. of water in the casing. If you have 100 ft. of water in the casing, you will use 0.018 L x 100 ft. = 1.8 L of 12% chlorine.
Using Table 1, calculate the amount of chlorine you will need for your well.
Casing diameter_______ Chlorine strength_______ L needed per 1 ft. of water_______ x _______ ft. of water in casing = _______ L of chlorine.
| If your well is located in a pit, proper safety precautions need to be taken during the chlorination procedure. It is best to use a licensed water well contractor who has the proper equipment and experience to do the job safely. Well pits are no longer legal to construct. |
Step 4
Siphon or drain this solution slowly into the well (see Figure 2, Siphoning Chlorine Solution).
Step 5
Open each hydrant and faucet in the distribution system (including all appliances that use water such as dishwasher, washing machine, furnace humidifier) until the water coming out has a chlorine odour. This will ensure all the plumbing fixtures are chlorinated. Allow the hot water tank to fill completely. Consult your water treatment equipment supplier to find out if any part of your water treatment system should be bypassed, to prevent damage.
Step 6
Leave the chlorine solution in the well and distribution system for 8 to 48 hours. The longer the contact time, the better the results.
Step 7
Open an outside tap and allow the water to run until the chlorine odour is greatly reduced. Make sure to direct the water away from sensitive plants or landscaping. Do not over-pump your well.
Step 8
Flush the chlorine solution from the hot water heater and household distribution system. The small amount of chlorine in the distribution system will not harm the septic tank.
Step 9
Backwash and regenerate any water treatment equipment. If you have an old well that has not been routinely chlorinated, consider hiring a
licensed water well contractor to thoroughly clean the well prior to chlorinating. Any floating debris should be removed from the well and the casing should be scrubbed to disturb the sludge buildup.
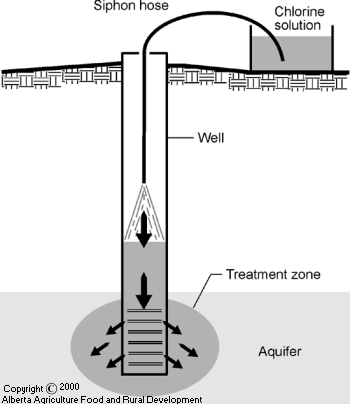
Figure 2 Siphoning Chlorine Solution
Modified Procedure for Large Diameter Wells
Due to the large volume of water in many bored wells the above procedure can be impractical. A more practical way to shock chlorinate a bored well is to mix the recommended amount of chlorine right in the well. An extra volume of chlorinated water is used to force some of the chlorine solution into the formation around the well. Follow these steps to shock chlorinate a large diameter bored well.
Step 1
Pump 200 gal. (1000 L) of water into a clean storage tank at the well head.
Step 2
Mix 4.0 L of 5 1/4% domestic chlorine bleach that does not have additives or perfumes (or 1.5 L of 12% bleach or 0.26 kg of 70% calcium hypochlorite) into the 200 gal. of stored water. This mixture will be used later in Step 5.
Step 3
Using Table 1 calculate the amount of chlorine you require per foot of water in the casing and add directly into the well. (Note that the 70% hypochlorite powder should be completely dissolved in a small container of water to form a solution before placing in the well.)
Step 4
Circulate the chlorinated water in the well by hooking a cleangarden hose up to an outside faucet and placing the other end back down the well. This circulates the chlorinated water through the pressure system and back down the well. Continue for at least 15 minutes.
Step 5
Siphon or drain the 200 gal. bleach and water solution prepared in Steps 1 and 2 slowly into the well.
Step 6
Complete the procedure as described in Steps 5 to 9 for drilled wells.
| Don't mix acids with chlorine. This is dangerous. |
|
|