| | Introduction | Materials and methods | Results | Conclusion | Barley energy calculator
Introduction
Barley is the most extensively used feed grain in western Canada. Feeding value depends on its protein and energy contents. Amino acids (AAs) are important to nutritionists when formulating diets for monogastric animals. These animals have a dietary requirement for certain essential AAs, some of which are in limiting supply in grains thereby affecting animal performance. Therefore, good data on the AA content of feed barley is important in order to accurately formulate diets for swine and poultry.
A knowledge of the AA content of barley is also important to cereal breeders and agronomy researchers. A quick and cheap way to obtain these analyses can speed up line selections for certain AA. Also the effect of various agronomic treatments on AA content can be studied. However, AA analysis is not readily available from local laboratories mainly because of the high cost which leads to low demand. Near Infrared Reflectance Spectroscopy (NIRS) offers the best possibility of lowering analysis cost and providing quick and reliable analyses. Previous studies have demonstrated that NIRS can be used to measure AA in ground grains but there is a strong industry need to determine whether this can be done for whole grains.
This study was carried out to determine if the NIRS system 6500 could be calibrated to accurately measure amino acids in whole barley grains, and to develop regression equations to calculate amino acids from dry matter crude protein.
Materials and Methods
For this study, 832 samples of hulled barley and 603 samples of hulless barley were used. These samples were selected over a five year period from several thousand research and farmer samples because of their diverse infra-red spectral profile.
Samples were scanned at the Soil and Crop Diagnostic Centre (SCDC) laboratory in Edmonton using a NIRSystems 6500. The CENTER and SELECT programs were used to order the samples according to spectra and select a symmetrically representative set of 150 samples from each type of barley. Amino acid analysis of the samples for 17 amino acids was performed at SCDC using Waters HPLC equipment and a new derivatization agent (AQC) which eliminates potential sources of laboratory error common with other derivatization agents.
Chemical analysis data of amino acids were converted to dry matter basis and used in the calibration program to prepare a calibration file. This procedure was performed separately for hulled and hulless barley. The cross validation method was used to generate prediction equations. The regression method used in the calibration procedure was a modified Principal Least Squares (MPLS) using 3, 5, 5, 1 as a math treatment and 248 wavelengths were chosen from 400 to 2498 nm. Detrend was used to reduce the interference of light scatter and particle size of sample in the spectra. Downweight was used to remove samples with large "T" or "H" values. SAS was used to calculate linear regression equations for amino acid on protein.
Results
Table 1 shows the mean contents of crude protein (N x 6.25) and seventeen AA in hulled and hulless barley on dry matter basis. Except for aspartic acid, hulless barley has higher AA levels than hulled barley. The higher levels are likely a reflection of higher average CP in hulless barley because of the lack of hulls that account for about ten percent of the weight of the kernel. Amino acid content of the protein fractions were significantly different (P<0.05) between hulled and hulless barleys for 14 of the 17 amino acids tested as shown in Table 2.
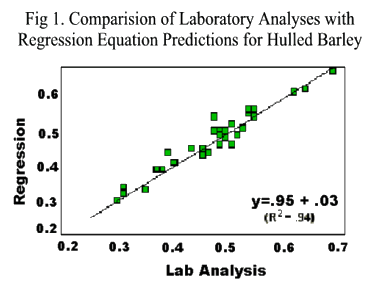
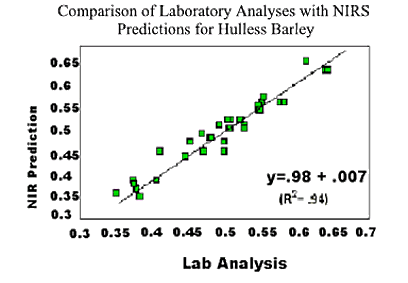
The statistical parameters of the NIRS prediction equations for amino acids in hulled and hulless barleys are shown in Tables 4 and 5 respectively. Except for glutamic acid, SEC and SECV were low for all AA. R2 for all AA were >0.9 demonstrating that prediction equations for whole barley were as accurate as for ground barley.
Linear regression equations for calculating AA in hulled and hulless barleys are shown in Table 3. R2 values of 0.87 to 0.98 indicate the high quality of these equations. Regression coefficients at the quadratic level were not significant. The combined chemical analysis data for AA from hulled and hulless barley were used to develop regression equations also shown in Table 3. These equations may be useful in calculating AA for mixtures of hulled and hulless barley. Their R2 values ranged from 0.73 to 0.97 which indicate a reasonable level of accuracy across AA.
Conclusions
On average, hulless barley is higher in protein than hulled barley.
2) Significant differences in amino acid content of protein in hulled and hulless barley were found in 14 of the 17 amino acid tested. Hulled barley protein is slightly higher in lysine than hulless barley.
3) NIR calibration equations developed for prediction of amino acids in whole barley grain are as precise as those developed for ground grain on the NIRS 6500 system
4) Protein is a good indicator of amino acid content of barley because of a relatively straight line relationship.
5) Regression equations were developed which can be used to calculate amino acids in hulled and hulless barley from crude protein values.
Financial support from Alberta Pork, Alberta Barley Commission and Alberta Agricultural Research Institute is gratefully acknowledged. Many thanks to Brian Rossnagel (CDC, Saskatchewan), Jim Helm, Ross McKenzie and other researchers in the PID division of AAFRD for their contribution of samples. The Soils and Crops Diagnostic Centre of Alberta Agriculture Food and Rural Development, Edmonton, provided the laboratory space, analytical equipment, the NIRSystems 6500 and the technical expertise that were necessary for this study. Their services are greatly appreciated.
Table 1. Protein and amino acid analyses (as % drymatter of 150 samples of hulled and 150 samples of hulless barleys grown in Alberta and Saskatchewan from 1990-1995.
| Amino Acid | Hulled Barley | Hulless Barley |
 | Mean | STD | Range | Mean | STD | Range |
| Protein | 13.87 | 2.89 | 7.82-22.7 | 15.91 | 3.68 | 8.48-22.6 |
| Alanine | 0.533 | 0.1 | 0.32-0.81 | 0.569 | 0.12 | 0.33-0.88 |
| Arginine | 0.638 | 0.148 | 0.34-1.11 | 0.727 | 0.15 | 0.39-1.02 |
| Aspartic acid | 0.813 | 0.158 | 0.48-1.31 | 0.774 | 0.142 | 0.46-1.11 |
| Cystine | 0.293 | 0.05 | 0.18-0.43 | 0.322 | 0.05 | 0.20-0.42 |
| Glutamic acid | 3.169 | 0.953 | 0.96-6.20 | 3.717 | 1.027 | 1.19-5.60 |
| Glycine | 0.552 | 0.1 | 0.34-0.85 | 0.599 | 0.103 | 0.39-0.78 |
| Histidine | 0.29 | 0.07 | 0.15-0.48 | 0.389 | 0.076 | 0.23-0.54 |
| Isoleucine | 0.458 | 0.11 | 0.22-0.79 | 0.559 | 0.133 | 0.31-0.81 |
| Leucine | 0.906 | 0.208 | 0.44-1.50 | 1.029 | 0.219 | 0.58-1.43 |
| Lysine | 0.471 | 0.09 | 0.27-0.76 | 0.507 | 0.095 | 0.31-0.73 |
| Methionine | 0.214 | 0.04 | 0.14-0.35 | 0.255 | 0.053 | 0.15-0.37 |
| Phenylalanine | 0.649 | 0.185 | 0.22-1.17 | 0.794 | 0.211 | 0.38-1.22 |
| Proline | 1.448 | 0.422 | 0.40-2.55 | 1.732 | 0.509 | 0.70-2.68 |
| Serine | 0.57 | 0.124 | 0.31-0.91 | 0.614 | 0.143 | 0.32-0.87 |
| Threonine | 0.449 | 0.09 | 0.25-0.69 | 0.488 | 0.09 | 0.30-0.66 |
| Tyrosine | 0.289 | 0.09 | 0.10-0.52 | 0.305 | 0.083 | 0.11-0.48 |
| Valine | 0.65 | 0.143 | 0.34-1.05 | 0.773 | 0.163 | 0.44-1.07 |
Table 2. Amino acid analyses (as % crude protein) of 150 samples of hulled and 150 samples of hulless barleys grown in Alberta and Saskatchewan from 1990-1995.
| Amino Acid | Hulled Barley | Hulless Barley |
 | Mean | STD | Range | Mean | STD | Range |
| Protein | 13.87 | 2.89 | 7.82-22.7 | 15.91 | 3.68 | 8.48-22.6 |
| Alanine | 3.88 | 0.25 | 3.29-5.24 | 3.61 | 0.24 | 3.14-4.39 |
| Arginine | 4.58 | 0.27 | 3.84-5.27 | 4.6 | 0.23 | 4.13-5.21 |
| Aspartic acid | 5.9 | 0.43 | 4.70-7.09 | 4.94 | 0.39 | 4.17-6.23 |
| Cystine | 2.14 | 0.17 | 1.74-2.68 | 2.07 | 0.22 | 1.69-2.60 |
| Glutamic acid | 22.6 | 2.53 | 14.1-27.7 | 23.1 | 1.69 | 16.7-25.5 |
| Glycine | 4.01 | 0.24 | 3.23-5.12 | 3.83 | 0.3 | 3.29-4.85 |
| Histidine | 2.09 | 0.18 | 1.33-2.62 | 2.47 | 0.15 | 2.15-2.96 |
| Isoleucine | 3.28 | 0.23 | 2.61-3.84 | 3.52 | 0.15 | 3.06-4.12 |
| Leucine | 6.51 | 0.4 | 5.45-7.52 | 6.5 | 0.25 | 5.85-7.57 |
| Lysine | 3.42 | 0.21 | 2.84-4.01 | 3.24 | 0.27 | 2.79-4.13 |
| Methionine | 1.56 | 0.1 | 1.28-1.85 | 1.61 | 0.1 | 1.39-1.92 |
| Phenylalanine | 4.61 | 0.52 | 2.81-6.02 | 4.95 | 0.26 | 4.29-5.57 |
| Proline | 10.25 | 1.18 | 5.12-12.62 | 10.7 | 0.91 | 8.25-12.2 |
| Serine | 4.11 | 0.23 | 3.40-4.79 | 3.86 | 0.13 | 3.53-4.24 |
| Threonine | 3.24 | 0.18 | 2.61-3.97 | 3.11 | 0.21 | 2.76-3.72 |
| Tyrosine | 2.05 | 0.26 | 1.28-2.55 | 1.9 | 0.19 | 1.10-2.33 |
| Valine | 4.68 | 0.26 | 3.97-5.35 | 4.89 | 0.22 | 4.32-5.94 |
Table 3. NIR regression statistics for whole and ground hulled barley grain using 120 calibration and 30 validation samples.
| Amino Acid | Whole | Ground |
| SEP | R2 | RPD | SEP | R2 | RPD |
| Alanine | 0.03 | 0.93 | 3.33 | 0.02 | 0.94 | 5.00 |
| Arginine | 0.03 | 0.94 | 5.00 | 0.03 | 0.95 | 5.00 |
| Aspartic acid | 0.05 | 0.91 | 3.20 | 0.04 | 0.95 | 4.00 |
| Cystine | 0.01 | 0.89 | 4.00 | 0.01 | 0.93 | 4.00 |
| Glutamic acid | 0.24 | 0.94 | 3.88 | 0.16 | 0.97 | 5.81 |
| Glycine | 0.02 | 0.94 | 5.00 | 0.02 | 0.96 | 5.00 |
| Histidine | 0.02 | 0.86 | 3.50 | 0.02 | 0.98 | 3.50 |
| Isoleucine | 0.03 | 0.93 | 3.67 | 0.03 | 0.97 | 3.67 |
| Leucine | 0.05 | 0.95 | 4.20 | 0.04 | 0.97 | 5.25 |
| Lysine | 0.03 | 0.92 | 3.00 | 0.02 | 0.94 | 4.50 |
| Methionine | 0.01 | 0.91 | 4.00 | 0.01 | 0.95 | 4.00 |
| Phenylala-nine | 0.04 | 0.96 | 4.75 | 0.04 | 0.96 | 4.75 |
| Proline | 0.11 | 0.94 | 3.91 | 0.07 | 0.97 | 6.14 |
| Serine | 0.04 | 0.93 | 3.25 | 0.03 | 0.94 | 4.33 |
| Threonine | 0.03 | 0.92 | 3.33 | 0.03 | 0.94 | 3.33 |
| Tyrosine | 0.03 | 0.91 | 3.00 | 0.03 | 0.92 | 3.00 |
| Valine | 0.04 | 0.93 | 3.50 | 0.03 | 0.98 | 4.67 |
SEP: Standard error of prediction
R2: Correlation coefficient between the HPLC analysed values and NIRS predicted results.
RPD: The ratio of standard deviation (SD) from the HPLC values divided by SEP.
Table 4. NIR regression statistics for whole and ground hulless barley grain using 120 calibration and 30 validation samples.
| Amino Acid | Whole | Ground |
| SEP | R2 | RPD | SEP | R2 | RPD |
| Alanine | 0.04 | 0.90 | 2.50 | 0.02 | 0.96 | 5.00 |
| Arginine | 0.04 | 0.93 | 3.25 | 0.02 | 0.98 | 6.50 |
| Aspartic acid | 0.04 | 0.89 | 3.00 | 0.03 | 0.94 | 4.00 |
| Cystine | 0.03 | 0.89 | 1.67 | 0.01 | 0.91 | 5.00 |
| Glutamic acid | 0.46 | 0.96 | 2.00 | 0.10 | 0.99 | 9.20 |
| Glycine | 0.03 | 0.91 | 3.00 | 0.02 | 0.96 | 4.50 |
| Histidine | 0.02 | 0.91 | 3.50 | 0.02 | 0.95 | 3.50 |
| Isoleucine | 0.04 | 0.94 | 3.00 | 0.01 | 0.99 | 12.00 |
| Leucine | 0.05 | 0.95 | 4.00 | 0.03 | 0.98 | 6.67 |
| Lysine | 0.03 | 0.91 | 2.67 | 0.02 | 0.93 | 4.00 |
| Methionine | 0.03 | 0.93 | 1.67 | 0.01 | 0.97 | 5.00 |
| Phenylala-nine | 0.08 | 0.94 | 2.25 | 0.02 | 0.99 | 9.00 |
| Proline | 0.11 | 0.95 | 4.00 | 0.05 | 0.99 | 8.80 |
| Serine | 0.07 | 0.92 | 1.86 | 0.02 | 0.97 | 6.50 |
| Threonine | 0.02 | 0.93 | 4.00 | 0.02 | 0.96 | 4.00 |
| Tyrosine | 0.03 | 0.89 | 2.67 | 0.02 | 0.94 | 4.00 |
| Valine | 0.04 | 0.95 | 3.75 | 0.02 | 0.98 | 7.50 |
SEP: Standard error of prediction
R2: Correlation coefficient between the HPLC analysed values and NIRS predicted results.
RPD: The ratio of standard deviation (SD) from the HPLC values divided by SEP.
Table 5. Regression equations for predicting % amino acid from dry matter crude protein % in hulled and hulless barleya.
[Amino Acid (%) = Intercept + (Slope X % crude protein, DM basis)]
 | Hulled barley | Hulless barley | Hulled and Hulless barley |
| Amino Acid | Inter-cept | Slope | SEb | R2 | Inter-cept | Slope | SEb | R2 | Inter-cept | Slope | SEb | R2 |
| Alanine | 0.092 | 0.032 | 0.027 | 0.92 | 0.063 | 0.032 | 0.028 | 0.95 | 0.096 | 0.031 | 0.031 | 0.92 |
| Arginine | -0.051 | 0.050 | 0.036 | 0.94 | 0.088 | 0.040 | 0.029 | 0.96 | 0.031 | 0.044 | 0.036 | 0.95 |
| Aspartic acid | 0.101 | 0.051 | 0.055 | 0.88 | 0.180 | 0.037 | 0.037 | 0.93 | 0.239 | 0.037 | 0.08 | 0.73 |
| Cystine | 0.087 | 0.015 | 0.014 | 0.91 | 0.116 | 0.013 | 0.016 | 0.90 | 0.103 | 0.014 | 0.015 | 0.91 |
| Glutamic acid | -1.187 | 0.316 | 0.224 | 0.94 | -0.684 | 0.277 | 0.143 | 0.98 | -0.844 | 0.289 | 0.201 | 0.96 |
| Glycine | 0.105 | 0.032 | 0.026 | 0.93 | 0.164 | 0.027 | 0.020 | 0.96 | 0.148 | 0.029 | 0.026 | 0.94 |
| Histidine | -0.006 | 0.021 | 0.024 | 0.87 | 0.068 | 0.020 | 0.017 | 0.95 | -0.004 | 0.023 | 0.034 | 0.84 |
| Isoleucine | -0.056 | 0.037 | 0.027 | 0.94 | -0.007 | 0.036 | 0.022 | 0.97 | -0.047 | 0.037 | 0.028 | 0.95 |
| Leucine | -0.061 | 0.070 | 0.051 | 0.94 | 0.092 | 0.059 | 0.033 | 0.98 | 0.033 | 0.063 | 0.046 | 0.96 |
| Lysine | 0.060 | 0.030 | 0.026 | 0.92 | 0.108 | 0.025 | 0.023 | 0.94 | 0.102 | 0.026 | 0.027 | 0.92 |
| Methionine | 0.031 | 0.013 | 0.011 | 0.93 | 0.031 | 0.014 | 0.012 | 0.95 | 0.022 | 0.014 | 0.013 | 0.94 |
| Phenylalanine | -0.209 | 0.062 | 0.050 | 0.93 | -0.112 | 0.057 | 0.028 | 0.98 | -0.170 | 0.060 | 0.042 | 0.96 |
| Proline | -0.525 | 0.142 | 0.098 | 0.95 | -0.448 | 0.137 | 0.073 | 0.98 | -0.480 | 0.139 | 0.086 | 0.97 |
| Serine | -0.004 | 0.041 | 0.034 | 0.92 | 0.001 | 0.039 | 0.020 | 0.98 | 0.025 | 0.038 | 0.033 | 0.94 |
| Threonine | 0.024 | 0.031 | 0.025 | 0.93 | 0.107 | 0.024 | 0.020 | 0.95 | 0.084 | 0.026 | 0.026 | 0.92 |
| Tyrosine | -0.115 | 0.029 | 0.025 | 0.92 | -0.037 | 0.022 | 0.026 | 0.91 | -0.046 | 0.023 | 0.032 | 0.86 |
| Valine | -0.016 | 0.048 | 0.035 | 0.94 | 0.074 | 0.044 | 0.025 | 0.98 | 0.015 | 0.047 | 0.034 | 0.96 |
a: P<0.0001
b: Std error of Y estimate
S. Jaikaran1, H. Hsu2, E. A. Prommer2 and G. Recinos Diaz2.
1Animal Industry Division and 2Plant Industry Division, Alberta Agriculture Food and Rural Development,
905 OS Longman Building,
Edmonton, AB, Canada,
T6H 4P2. |
|