| | Background | Factsheet topics | Disease cycle | Clubroot symptoms on canola and mustard | Prevention and management of clubroot in canola and mustard | E-book
Clubroot is a serious soil-borne disease of cruciferous crops (canola and cabbage family) worldwide and was first identified in Europe in the thirteenth century. This disease is a major problem in cole crops (cruciferous vegetables) in some areas of British Columbia, Quebec, Ontario and the Atlantic provinces.
Background
There have been two previous reports of clubroot in cole crops in Alberta. Thus, clubroot is not a new disease in Canada or Alberta. However, in 2003, clubroot was confirmed in several canola fields near Edmonton, Alberta, which was the first report on canola in western Canada.
Clubroot has continued to spread in central Alberta. An Alberta map showing the current clubroot status by county can be found at: www.agriculture.alberta.ca
Clubroot was added as a declared pest to Alberta’s Agricultural Pests Act (APA) in April 2007. The APA is the legislative authority for the enforcement of control measures for declared pests in Alberta. The Alberta Minister of Agriculture and Rural Development is responsible for this Act.
However, enforcement of pest control measures is the responsibility of the municipal authority, and Agricultural Fieldmen are responsible for enforcing pest control measures in their municipality. Pest inspectors have the power to enter land at a reasonable hour, without permission, to inspect for clubroot and collect samples. The owner or occupant of land has the responsibility for taking measures to prevent the establishment and spread of clubroot.
Clubroot can affect broccoli, Brussels sprouts, cabbage, cauliflower, Chinese cabbage, kale, kohlrabi, radish, rutabaga and turnip. Canola/ rapeseed and mustard are also susceptible to this disease. Cruciferous weeds are susceptible as well. There are several weak, non-cruciferous hosts, but their contribution to disease development and carryover of the clubroot pathogen is not well understood.
This factsheet contains current information about clubroot in canola and describes options for Canadian canola growers to prevent this disease from being introduced and becoming well established in their fields.
Factsheet Topics
- disease cycle
- clubroot symptoms
- prevention and management
Disease Cycle
The causal agent of clubroot is Plasmodiophora brassicae Woronin. In the past, this agent has been classified as a slime mould fungus (myxomycete), but more recently, it is regarded as a protist (an organism with plant, animal and fungal characteristics).
There are normally several different races or pathotypes present in established infestations. Plasmodiophora brassicae is an obligate parasite, which means the pathogen cannot grow and multiply without a living host. The life cycle of P. brassicae is shown in Figure 1.
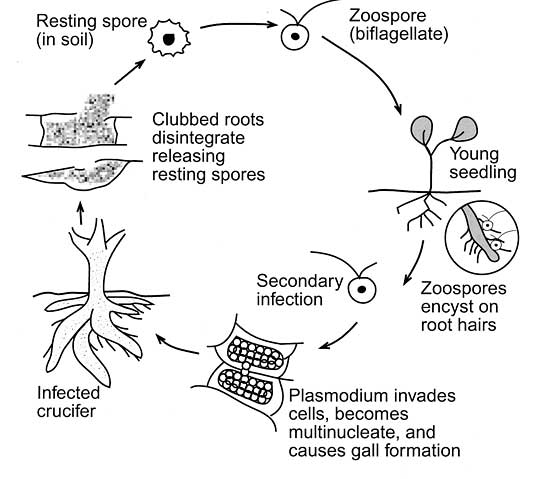
Figure 1. Life cycle of Plasmodiophora brassicae, the pathogen that causes clubroot.(Source: Ohio State University).
Resting spores germinate in the spring, producing zoospores that swim very short distances in soil water to root hairs. These resting spores are extremely long lived, with a half-life of about 4 years, but they can survive insoil for up to 20 years. For example, Swedish research in clubroot-infested spring rapeseed fields found that17 years were needed to reduce the infestation tonon-detectable limits.
The longevity of the resting spores is a key factor contributing to the seriousness of the disease. Resting spore germination is stimulated by exudates from the roots of host plants.
After the initial infection through root hairs or wounds, the pathogen forms an amoeba-like cell. This unusual cell multiplies and then joins with others to form a plasmodium, which is a naked mass of protoplasm with many nuclei. The plasmodium eventually divides to form many secondary zoospores that are released into the soil.
These second-generation zoospores re-infect roots of the initial host or nearby plants and are able to invade the cortex (interior) of the root. Once in the cortex, the amoeba-like cells multiply or join with others to form a secondary plasmodium. As this plasmodium develops, plant hormones are altered, which causes the infected cortical cells to swell. Clusters of these enlarged cells form “clubs” or galls (see Figures 2, 3 and 4).

Figure 2. Very severe clubroot on canola.
Photo credit: Kelly Turkington

Figure 3. Severe clubroot galls or “clubs” on canola root.
Photo credit: Kelly Turkington

Figure 4. Moderately infected canola root.
Photo credit: Valerie Sowiak
Some amoeba-like cells are able to move up and down roots in vascular tissue. After the secondary plasmodia mature, they divide into many resting spores within the gall tissue. The galls are quickly decayed by soil microbes, leaving millions of resting spores in the soil.
Although there are no airborne spores released by this pathogen, the resting spores are capable of moving with infested soil transported by wind or water erosion and field machinery.
Warm soil (20-24�C), high soil moisture and acid soil(pH less than 6.5) are environmental factors that favour infection and severe disease development. Unfortunately, these conditions exist in a significant portion of the traditional canola growing areas of Alberta.
High soil moisture areas of the field typically have the most severe infestations. These wet areas are found in depressions, spots with higher clay content or with subsoil horizons that cause poor water infiltration (such asGray Wooded or solonetzic soils).
Clubroot Symptoms on Canola and Mustard
Clubroot galls are a nutrient sink, so they tie up nutrients, and severely infected roots of canola cannot transport sufficient water and nutrients for aboveground plant parts. Symptoms will vary depending on the growth stage of the crop when infection occurs. Early infection at the seedling stage can result in wilting, stunting and yellowing of canola plants in the late rosette to early podding stage.
Such symptoms may be wrongly attributed to heat stress during periods with high temperatures or to other diseases such as blackleg or Fusarium wilt. In such cases, proper diagnosis includes digging up wilted plants to check for gall formation on roots.
Infection that occurs at later stages may not show plant wilting, stunting or yellowing. However, infected plants will ripen prematurely, and seeds will shrivel. Thus, yield and quality (oil content) are reduced.
Swedish researchers found that infestations nearing100 per cent-affected plants caused about 50 per centyield loss, while infestations of 10 to 20 per cent led to5 to 10 per cent yield loss. This result is similar to sclerotinia stem rot infection in canola, where a general rule of thumb yield loss estimate is half of the percentage of infected stems. This is a reasonable comparison since both diseases restrict the flow of water and nutrients to developing seeds.
Patches of prematurely ripening canola due to clubroot infection (Figure 5) could be confused with other diseases such as sclerotinia, blackleg and Fusarium wilt. In such cases, proper diagnosis should include digging up affected plants to check for gall formation on roots. Swathing is an excellent opportunity to spot clubroot infestations.

Figure 5. Patchy, premature ripening.
Photo credit: Stephen Strelkov
If the suspected plants are not sampled until after swathing, root galls may have decayed already, and the typical whitish galls will no longer be present (seeFigure 6). Instead, decayed root galls have a brown peaty appearance rather than the healthy white colour of unaffected root tissue, which should be a signal to carefully dig up more roots for closer inspection.

Figure 6. Decayed clubroot galls and whitish stem appearance.
Photo credit: Kelly Turkington
Hybridization nodules on canola roots (see Figure 7), although rare, could be confused with clubroot galls, but they appear as small, round nodules located at root nodes. The interior texture of a clubroot gall is spongy or marbled while hybridization nodules are uniformly dense inside, like healthy roots. Furthermore, hybridization nodules will not decay rapidly and do not have a peaty appearance like clubroot galls do.

Figure 7. Hybridization nodules on canola root.
Photo credit: Alvin Eyolfson
Phenoxy damage to canola may also induce galls on stem bases and roots (Figure 8). As with hybridization nodules, the phenoxy-induced galls will not decay rapidly like clubroot galls. Thickened, corky or split stem bases and curving stems are additional phenoxy symptoms that often occur in canola.

Figure 8. Root galls on canola due to phenoxy injury.
For confirmation of suspected canola clubroot galls, send samples to one of the commercial seed testing labs in the province.
Prevention and Management of Clubroot in Canola and Mustard
Since clubroot infestations are still not widely distributed in Alberta, producers should take various precautionary measures to curb the spread of this disease outside the known infested areas.
Prevention
Recommended preventative measures include the following:
- Use long rotations – grow canola not more frequently than once every four years (i.e. three years out of canola). Although this practice will not prevent the introduction of clubroot to clean fields, it will restrict this and other canola disease development within the field and probably avert a severe infestation. This rotation recommendation is similar to Europe where clubroot is more established, and, thus, more experience has been gained in management of this disease (see the Scottish clubroot factsheet by S. Oxley, 2007, noted in the References section at the end of this factsheet).
- Planting clubroot-resistant varieties on fields without the disease can be useful when clubroot is present nearby. This strategy relies on the genetic resistance to greatly reduce disease development/establishment compared to susceptible varieties if clubroot is inadvertently introduced to the field.
- Practice good sanitation to restrict the movement of possibly contaminated material (this approach will help reduce the spread of other diseases, weeds and insects too). The resting spores are most likely to spread via contaminated soil and infected canola plant parts. Thus, producers should follow the practice of cleaning soil and crop debris from field equipment before entering or leaving all fields. The equipment cleaning procedure involves knocking or scraping off soil lumps and sweeping off loose soil.
For risk-averse producers, the following additional cleaning steps may provide some extra benefit but involve considerably more work and expense:
- After removal of soil lumps, wash off equipment with a power washer.
- Finish by misting equipment with weak disinfectant (1-2% household bleach solution or EcoClear or HyperOx). The use of a disinfectant without first removing soil is not recommended because soil deactivates most disinfectants. The disinfectant must remain wet for 20 to 30 minutes on the equipment.
- Use direct seeding and other soil conservation practices to reduce erosion. Resting spores move readily in soil transported by wind or water erosion and overland flow.
- Scout canola fields regularly and carefully. Identify causes of wilting, stunting, yellowing and premature ripening – do not assume anything!
- Avoid the use of straw bales and manure from infested or suspicious areas. Clubroot spores are reported to survive through the digestive tracts of livestock.
- Avoid common untreated seed (including canola, cereals and pulses). Earth-tag on seed from infested fields could introduce resting spores to clean fields. Certain seed treatment fungicides may control spores on contaminated seed, but this observation needs further research to confirm.
Note: the risk of spreading clubroot by contaminated seed or straw is much lower than by the transportation of soil and plant debris on contaminated field equipment.
Management
Managing clubroot after establishment in a canola field is difficult and long term.
There are clubroot-resistant Canadian canola varieties available now. However, clubroot resistance in European varieties has not been durable there, and since 2014, resistance in Canadian canola varieties has started to be overcome by several new pathotypes in central Alberta. Canola growers in high risk situations (confirmed clubroot in the field or area) should follow traditional canola rotation recommendations (one canola crop every four years) using clubroot resistant varieties. The one in four year rotation recommendation using resistant varieties is designed to slow down pathogen population shifts to strains not controlled by current resistant varieties and allow time for new resistance sources to be bred into canola.
Resistance breakdown is not a change in the plant, but rather in the clubroot pathogen where it adapts to the resistance in the varieties being grown. There are several prevalent clubroot race(s) or pathotypes in the Alberta infestations, with one pathotype dominating. However, characterization of single spore-derived isolates from the populations indicates there are additional pathotypes and mixtures of pathotypes.
Clubroot galls may still be found at low levels in fields seeded to resistant varieties! Under high pressure, small galls may develop on plants with clubroot resistance. Galls can also occur on volunteers from previous canola crops of susceptible varieties, mustard family weed species and in the small percentage of off-types in hybrid seed (usually non-hybrid parental lines where one parental line may be clubroot susceptible).
Currently, there are no registered fungicides for clubroot control or suppression in canola. Although there are fungicides registered for clubroot control in cole crops around the world, the relatively high cost and application method (transplant bed drench or broadcast incorporation) make them unsuitable for canola field production.
Liming acid soils to above pH 7.2 has shown poor or erratic results for clubroot control in cole crops in British Columbia and eastern Canada. Given the inconsistency and high cost, liming is not a reliable option for clubroot control in canola.
Calcium cyanamide, an old form of nitrogen fertilizer with fungicidal properties, has shown promise for reducing clubroot in cole crops, but high application rates, significant cost and limited availability make it a poor option for canola.
Volunteer canola and susceptible weeds (mustard family, dock and hoary cress) must be controlled in the rotation crops. There is some evidence that a few non-cruciferous crops such as orchardgrass and red clover may be weak hosts for clubroot disease, but the rotational effect of such crops on clubroot incidence and severity is likely of little practical significance.
In combination with the rotation strategy, sanitation and soil conservation measures should be practiced to keep contaminated soil and infected crop debris from being transported from infested fields. Whenever practical, infested fields should not be worked in when wet since more mud will stick to equipment and then be transported to clean fields.
There has been one report from Norway of lower clubroot severity under reduced tillage. Thus, reduced tillage or direct seeding may help to combat a clubroot infestation, and the fewer tillage operations will help to avoid the transport of contaminated soil. Similarly, all equipment traffic into infested fields should be minimized – for example, service and nurse trucks should remain on the road and field equipment be brought to them.
Clubroot disease is a serious concern in Alberta. Understanding the disease cycle, recognizing the symptoms and adopting good prevention and management practices can assist in its control.
References
Cao, T., Tewari, J.P., and Strelkov, S.E. 2007. Molecular detection of Plasmodiophora brassicae, causal agent of clubroot of crucifers, in plant and soil. Plant Dis. 91:80-87.
Ekeberg, E. and Riley, H.C.F. 1997. Tillage intensity effects on soil properties and crop yields in a long-term trial on morainic loam soil in southeast Norway. Soil & Tillage Res. 42: 277-293.
Friberg, H., Lagerlof, J., and Ramert, B. 2006. Usefulness of nonhost plants in managing Plasmodiophora brassicae. Plant Pathol. 55:690-695.
McDonald, M.R., Kornatowska, B. and McKeown, A.W. 2002. Management of clubroot of Asian Brassica crops grown on organic soils. Abstract S08-0-11, XXVIth International Horticulture Congress, Toronto.
Oxley, S. 2007. Clubroot disease of oilseed rape and other brassica crops. Technical Note 602. Scottish Agricultural College. Accessed on-line October 5, 2010. http://www.sruc.ac.uk/download/downloads/id/1220/tn602_clubroot_disease_of_oilseed_rape_and_other_brassica_crops
Strelkov, S.E., Tewari, J.P., and Smith-Degenhardt, E. 2006. Characterization of Plasmodiophora brassicae populations from Alberta, Canada. Can. J. Plant Pathol. 28:467-474.
Tewari, J.P., Strelkov, S. E., Orchard, D., Hartman, M., Lange, R., and Turkington, T.K. 2005. Identification of clubroot of crucifers on canola (Brassica napus) in Alberta. Can. J. Plant Pathol. 27:143-144.
Tremblay, N., B�lec, C., Lawrence, H., and Carisse, O. 1999. Clubroot of crucifers – control strategies. Agriculture and Agri-Food Canada. Saint-Jean-sur Richelieu, PQ. 3 pp.
Wallenhammar, A.C. 1996. Prevalence of Plasmodiophora brassicae in a spring oilseed rape growing area in central Sweden and factors influencing soil infestation levels. Plant Pathol. 45:710-719.
Wallenhammar, A.C., Johnsson, L., and Gerhardson, B. 1999. Clubroot resistance and yield loss in spring oilseed turnip rape and spring oilseed rape. Proceedings of 10th International Rapeseed Congress, Australia.
Prepared by
Murray Hartman, Oilseed Specialist, Agriculture and Rural Development
With contributions from the following plant pathologists: J.P. Tewari, Kelly Turkington, Ralph Lange, Stephen Strelkov, Ron Howard and Paul Laflamme
For more information
Alberta Ag-Info Centre
Call toll free: 310-FARM (3276)
Source: Agdex 140/638-1. Revised September 2015. |
|