| | Background | Effect of sulphur on crop growth | Sulphur deficiency symptoms | Indirect causes of sulphur deficiency | Soil testing for sulphate-sulphur | Application methods | Sulphur fertilizer recommendations
Sulphur (S) is an essential plant nutrient required by all crops for optimum production. Plants take up and use S in the sulphate (SO4-S) form, which like nitrate (NO3-N), is very mobile in the soil and is prone to leaching in wet soil conditions, particularly in sandy soils.
Sulphur deficiencies are becoming increasingly common in Alberta. Deficiencies can be easily corrected with fertilizers containing sulphate (S04). Generally, S is the third most limiting soil nutrient in cereal, oilseed and forage crop production in Alberta. It is third only to nitrogen (N) and phosphorus (P) in fertilizer use in Alberta.
Background
Oilseed crops, particularly canola, and forage crops, have a higher S requirement than cereal crops. Table 1 provides examples of nutrient uptake and removal by wheat, canola, pea and alfalfa. Sulphur is required in the development of fertile canola flowers and must be present for good nodule development on legume forages such as alfalfa and pulse crop roots such as pea and faba bean.
In Alberta, an estimated 6 to 8 million acres are considered potentially S deficient for optimum canola production, and the potentially deficient areas are increasing due to increased crop yields and increased canola production, which is drawing down S soil reserves.
Soil organic matter is the primary source of plant-available SO4-S in surface soil. Soils that are sandy, low in organic matter and found in upper to mid-slope field positions are particularly prone to S deficiency since only a small amount of SO4-S is released from organic matter and is susceptible to leaching loss.
The subsoil of Brown and Dark Brown soils in southern and south central Alberta often have an abundance of gypsum, which is calcium sulphate (CaSO4). This mineral is an important source of plant-available S in these soils.
| Table 1. Nutrient levels taken up and removed by average yields of canola, wheat and alfalfa in Alberta |
| Crop | Yield | Crop Part | Nitrogen
N | Phosphate
P205 | Potassium
K20 | Sulphur
S |
(lbs/acre) |
| Canola | 35 bu/ac | Seed | 60-75 | 30 - 35 | 15 - 20 | 10 - 12 |
| Seed/straw | 100 - 115 | 45 - 50 | 75 - 85 | 17 - 20 |
| Wheat | 50 bu/ac | Seed | 60 - 75 | 24 - 28 | 70 - 85 | 10 - 12 |
| Seed/straw | 85 - 110 | 32 - 36 | 15 - 22 | 5 - 6 |
| Pea | 50 bu/ac | Seed | 100 - 120 | 30 - 35 | 30 - 35 | 6 - 7 |
| Seed/straw | 130 - 150 | 35 - 45 | 120 - 140 | 10 - 14 |
| Alfalfa | 5 tons/ac | Total | 260 - 320 | 60 - 75 | 270 - 330 | 27 - 33 |
Soils most prone to S deficiency are the Thin Black, Black and Gray Wooded soils (Figure 1) because they were formed in areas of relatively high rainfall with greater leaching potential and have higher yield potential, meaning a higher S uptake by crops.
Brown and Dark Brown soils in southern and south central Alberta are usually not considered S deficient because they often have relatively high levels of gypsum in the subsoils. However, low levels of SO4-S may occur in the surface soil, even while the subsoil has adequate plant-available SO4-S levels.
For sensitive crops such as canola, sulphate fertilizer may be needed to ensure good vegetative growth earlier in the season, until plant roots have penetrated into the subsoil that has adequate S. Therefore, within all soil zones, various combinations of high rainfall, high yield potential, low organic matter, topography and coarse texture predispose fields to potential S deficiency. As such, producers must be aware of the potential soil conditions for sulphur deficiency on their land.
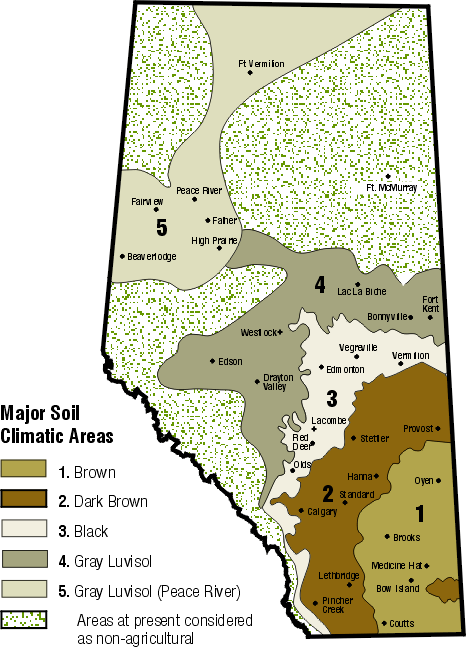
Figure 1. Soils map of Alberta
Effect of Sulphur on Crop Growth
Sulphur is essential in the structural and enzymatic components in plants. Sulphur is a key component of some essential amino acids and is needed for protein synthesis. Chlorophyll synthesis also requires S.
Sulphur is not readily translocated within plants, so all plants need a continuous supply of sulphur from emergence to crop maturity. Therefore, in S-deficient plants, older leaves may appear more healthy, while newer leaves and tissue may have stunted growth and a lighter green or even yellow appearance.
A sulphur deficiency at any growth stage can result in reduced crop growth and yield. Adequate S results in rapid crop growth and earlier maturity.
Sulphur Deficiency Symptoms
Sulphur deficiency symptoms vary between crops. In canola, deficiency symptoms may begin as early as the one-leaf stage (Figure 2a), with the newest leaves turning yellowish green with dark vein coloration. Leaves may take on a “cupped” appearance and later, a reddening from the leaf margins (Figures 2b and 2c).
Flowers are usually smaller and paler yellow, and they produce small, underdeveloped purplish-coloured pods (Figure 2d). Under mild S deficiency, there may be good vegetative growth, but flowers and pods will be underdeveloped.
For cereals and forage grasses, the yellowing of newly emerging leaves is a strong indicator of S deficiency (Figure 3). Depending on the degree of deficiency, the leaves may be a shade of light green to entirely yellow.
Yellowing of the new growth occurs because S is immobile in the plant; therefore, newly emerging leaves cannot “scavenge” S from older leaves. This situation is in contrast to nitrogen deficiency symptoms in which the older leaves turn yellow first because nitrogen is translocated or “scavenged” from older leaves to support new growth.
Figure 2. Sulphur deficiency symptoms in canola.

2a. Sulphur deficient seedling.

2b. Leaf cupping.

2c. Dark vein coloration and purpling of leaf margins.

2d. Small pale flowers and poor pod development.
Sulphur deficiency in alfalfa results in shorter plant stature with pale green to yellow leaves (Figure 4a and b). The yellowing is often, but not always, localized to newly emerging leaves. Sulphur deficiency can result in nitrogen deficiency in alfalfa since S is required for health and function of nitrogen-fixing nodules. Sulphur deficiency in alfalfa occurs more commonly and is more severe in Gray Wooded soil.

Figure 3. Yellowing of newly emerging leaves is an indicator of sulphur deficiency in wheat, other cereals and forage grasses.
Figure 4. Sulphur deficiency in alfalfa.

4a. Deficient alfalfa plant on left is shorter in stature with yellowing most severe on young leaves.

4b. In the field, sulphur-deficient alfalfa on the right.
Similar symptoms
Great care must be exercised when diagnosing nutrient deficiencies, and using visual symptoms alone can be very misleading. The diagnosis of problems must include using soil and tissue sampling and the analysis of affected and unaffected areas of a field to confirm a nutrient deficiency.
Sulphur deficiency can be similar to and confused with many other stressors that cause symptoms in canola similar to those caused by S deficiency. For example, Group 4 herbicides such as 2,4-D or dicamba (Banvel) at low, sub-lethal rates from wind drift, tank contamination or even precipitation can cause leaf cupping in canola (Figure 5a). However, the leaves in this figure lack the dark vein coloration associated with S deficiency.
Like S deficiency, Group 2 herbicides can cause purpling of canola leaves, but unlike the leaves of S-deficient canola, the purpling does not develop from the leaf margins.
Figure 5b shows Muster (ethametsulfuron-methyl) injury on canola. Muster is a registered herbicide (Group 2) on canola, but injury could occur if a heavy rainfall occurs shortly after spraying, facilitating the uptake of the herbicide via the roots.

Figure 5a. Canola leaf cupping from Banvel injury.

Figure 5b. Purpling of older leaves resulting from Muster sprayed just prior to heavy rainfall.
Other Group 2 herbicides such as Ally (metsulfuron-methyl) or Pursuit (imazethapyr) can carry-over in the soil and cause purpling of the older canola leaves. If Group 2 herbicides are sprayed directly on canola as a tank contaminant, purpling of the youngest leaves may occur.
Boron deficiency can produce thin purple pods in canola, which are hard to distinguish from S-deficiency symptoms. Fortunately, boron deficiencies in Alberta soils are rare. Sun scald produces purpling, but the discoloration is confined to the “sunny” side of stems and pods.
Indirect Causes of Sulphur Deficiency
Sulphur deficiency in canola can occur despite the adequate application of S fertilizer. Any stressor causing root pruning or restricted root growth can reduce the plant’s ability to take up nutrients, which can potentially induce S and other nutrient deficiencies.
Herbicide carry-over, acidic soil or a shallow hard pan soil layer can cause restricted root growth and restrict sulphate-S uptake, leading to deficiency symptoms. Root rot diseases, some of which are preventable with seed treatment, can cause the development of sulphur deficiency in canola (Figure 6).

Figure 6. Canola grown from bare seed (left) versus Vitavax-treated (right ).

Inset: Close-up of canola grown from bare seed showing sulphur deficiency resulting from seedling blight.
Soil Testing for Sulphate-Sulphur
Plant-available S in soil is determined by measuring the soluble sulphate-sulphur content. This soil test provides a reliable basis for making S fertilizer recommendations as long as representative soil samples can be obtained from the field.
Ideally, samples should be taken just before seeding. However, late fall sampling is usually satisfactory to provide reliable fertilizer recommendations when samples are taken after the surface soil temperature drops below 5� C (generally after mid-October).
A soil sampling probe should be used to obtain samples. Take soil samples from 0 to 6, 6 to 12 and 12 to 24 inches (0 - 15, 15 - 30 and 30 - 60 cm) depths. Fertilizer recommendations for phosphorus and potassium are based on the 0 to 6 inch depth.
Soil testing laboratories will provide fertilizer recommendations for S based on a 0 to 6 inch or 0 to 6 plus 6 to 12 inch depth; however, this approach may provide a higher than reasonable recommendation because available S in the subsoil is not taken into consideration. With each extra soil sample taken from a greater soil depth (6 - 12 inch, 12 - 24 inch), the fertilizer recommendations for S become more precise.
Even with proper sampling depth, obtaining a representative sample from the field can be a challenge. Studies across all soil zones have found the level of crop-available S can vary greatly within a field.
For example, grid sampling (0 - 60 cm) within a field near Stettler found sulphate levels varied from 9 to 6,330 ppm with the average being 588 ppm. Based on a random composite sample, no fertilizer recommendation for S would be made for this field. However, the mode (the value of most frequent occurrence) was 11 ppm. Based on this number, S fertilizer would be recommended. If the soil test results from the composite sample were followed, the majority of this field would be lacking adequate S. Obtaining a representative sample from fields highly variable in available S is difficult.
The benchmark or topographic system of soil sampling can help with fertilizer decisions. For benchmark sampling, the location selected must be representative of the field; if it is not representative, very misleading information could lead to over or under application of sulphur fertilizer.
With the benchmark system, several representative sites in a field should be selected, and each site should be about 100 feet by 100 feet (30 by 30 m). Each year, 15 to 20 sample cores are taken from each benchmark site. Provided the benchmark sites are representative, this system gives a good indication of the nutrient status of the field and how it varies from year-to-year.
Finding good benchmark sites may be difficult. However as a general rule, top slope and sandy soils are often associated with S deficiency and should be sampled separately from low relief or heavy textured soils.
Sulphur Fertilizer Types
Sulphur fertilizers are most commonly available in either the soluble sulphate form or elemental forms (S�), which are relatively insoluble. In Alberta, products containing ammonium sulphate (21-0-0-24; 20-0-0-24) are commonly used and are excellent on deficient soils. Other products such as potassium sulphate can be used to correct both potassium and sulphur deficiencies but this fertilizer product is not commonly available.
Products comprised of elemental So, such as 0-0-0-90 (Tiger 90CR) and 0-0-0-95 (SulFer 95), are becoming increasingly popular, but elemental products must be carefully managed to be effective. Other newer products contain both sulphate-sulphur and elemental S such at the MicroEssentials� S15™ (13-33-0-15S).
Provided there is adequate soil moisture, ammonium sulphate fertilizers dissolve quickly to release SO4-S, the sulphate form readily available to plants. As mentioned, SO4-S is quite mobile in the soil and is the only form of S directly utilized by plants. Plants cannot take up elemental S. These fertilizers must be converted to sulphate-S first before plant uptake can occur.
Application Methods
Because ammonium sulphate fertilizers are so rapidly available to crops, they can be applied to crops in various ways. Normally, the uptake efficiency of soluble sulphate fertilizer is better when banded in soil versus broadcast and incorporation.
When sulphate-S is sided banded near the seed or seed-placed, uptake efficiency tends to be best. Further, banded S fertilizer may favour crop growth versus weed growth, since this method places the fertilizer close to the crop roots.
Normally broadcast application onto snow is not recommended. Some sulphate loss due to redistribution in runoff may occur if ammonium sulphate fertilizers are broadcast on snow deeper than 4 inches (10 cm) covering frozen ground, particularly on slopes greater the 4 per cent.
If detected early enough, S deficiency can be corrected in many crops by broadcasting ammonium sulphate. In the case of canola, broadcasting ammonium sulphate until the early flowering stage may help correct an S deficiency provided there is enough rain to dissolve and move the SO4-S into the root zone (Figure 7). The greatest risk from using soluble sulphate fertilizers occurs on sandy soils subjected to heavy rainfall. Under these conditions, SO4-S may leach below the rooting zone.

Figure 7. Canola on the left was rescued from sulphur deficiency by applying 100 lbs/ac of 20-0-0-24 applied at early flower.
For fertilizer manufacturers, elemental sulphur (S�) has the advantages of lower production costs and because of its high analysis, lower transportation costs. In addition, the application of S� does not add any extra nitrogen, which could reduce yields of legumes such as alfalfa or pulse crops such as peas. High soil nitrogen can reduce legume yields by interfering with proper nodule formation and nitrogen fixation.
Elemental S� fertilizer must be oxidized by soil microbes to SO4-S before it is available to crops. Typically, it takes considerably more time for S� to become available compared to soluble sulphate forms of fertilizer. The rate of conversion from S� to plant-available SO4-S depends on two factors: the particle size to which the product degrades and the method of application.
Alberta research has determined that S� granules that break down into particles smaller than 150 Microns (10-6 m) convert more quickly to sulphate, since particles of this size or smaller provide sufficient surface area for microbes to act on and solubilize S. After two simulated wet/freeze cycles and one rainfall, SulFer 95 (0-0-0-95) particles are all <74 microns whereas only 25 per cent of Tiger 90 particles were <150 microns.
The evolution of elemental S� products has been rapid, and a number of elemental S� fertilizers are now available. Industry is continually striving to improve the dispersing nature of their S� products, making it difficult to give current comparisons between these various S� products.
For best management, regardless of brand, all S� products should be surface broadcast without incorporation at least a year before benefits are needed. Elemental S� placed on the soil surface will easily disperse on the surface and is more easily oxidized by soil microbes because rainfall and freeze/thaw cycles break down and disperse the S� granules.
Banding or immediate broadcasting followed by incorporation is an inefficient method of applying S� and is not recommended. Banding in soil or incorporating S� immediately after broadcasting will prevent S� granules from breaking down and dispersing. The surrounding soil would keep the S� granule intact, reducing the exposed S� surface area and vastly reducing the conversion rate to SO4-S. This situation would greatly reduce the microbial oxidation process and slow the release of S� to plant-available sulphate. Even with broadcast applications, the rate of conversion from S� to SO4-S can vary from less than a year to several years or more.
Producers must use care in deciding whether to use ammonium sulphate fertilizer versus elemental S�. Various Alberta studies have found canola yields to be significantly greater when fertilized with a spring-banded application of ammonium sulphate (20-0-0-24) compared to spring or even fall broadcast Tiger (Figure 8).
 |  |
Figure 8. Fall-applied Tiger 90 (left) versus spring-banded 20-0-0-24 (right).
Fertilizers containing sulphur as thiosulphate, such as liquid ammonium thiosulphate (12-0-0-26), must also be oxidized by microbes in the soil to convert to sulphate-S. Generally, the oxidation is somewhat more rapid than in granular form.
Livestock manure as S fertilizer
Livestock manure can provide S along with other plant nutrients. However, the sulphur content and balance with other nutrients can be variable. For example, some forms of manure may have low S relative to nitrogen (N). It has been observed that N:S ratios vary from 7:1 to 17:1 for cattle manure and from 13:1 to 25:1 for pig slurry.
N:S ratios greater than 10:1 may be too great for optimum canola production, which typically requires an N:S ratio of 7:1. Therefore, it is important to check the plant-available S levels in soil, and if the soil is deficient, supplemental S fertilization may be required when using animal manure to fertilize canola.
Irrigation water
The irrigation water in southern Alberta naturally contains sulphate-S. Approximately 30 lb/ac of sulphate-S is applied to soil in 12 inches (30 cm) of irrigation water. The amount of sulphate applied will vary within irrigation districts and time of year. The amount of S applied in irrigation water is normally more than sufficient to meet crop requirements over a growing season.
Sulphur Fertilizer Recommendations
As mentioned, sulphur deficiency in southern Alberta on Brown and Dark Brown soils and irrigated soils is less common. Deficiencies are more common, particularly with canola, on Thin Black, Black and Gray soils in central and northern Alberta.
A soil test for S can be very useful to determine if S fertilizer is required. Ensure that samples are taken separately from the 0 to 6, 6 to 12 and 12 to 24 inch depths to determine the level of S at various depths.
Table 2 provides a general guide to determine if S fertilizer is required and approximate rates of application. To ensure adequate S nutrition for optimum crop production, it is important to apply sulphate fertilizer, particularly in oilseed crops, when a surface soil is low in sulphate-sulphur.
| Table 2. General fertilizer recommendations for cereal, oilseed and pulse crops in Alberta |
| Soil test level S (lb/ac)z | S recommendation (lb/ac) |
| (0 to 6 + 6 to 12 inch depth) | Grains | Canola | Pulse |
| 30 | 0 | 0 | 0 |
| 20 - 30 | 0 | 10 | 5 |
| 15 - 20 | 5 | 15 | 10 |
| 10 - 15 | 10 | 20 | 15 |
| 5 - 10 | 15 | 25 | 20 |
| 0 - 5 | 20 | 30 | 25 |
z CaCl extraction method.
Sulphate-sulphur, like nitrate-nitrogen, is very mobile in soil. After heavy precipitation or irrigation events, sulphate leaching from the topsoil into subsoil can be a concern. Sandy soils have the greatest potential for sulphate leaching. After significant precipitation that can cause leaching, a deficiency in plant-available sulphate-sulphur may develop in surface soil.
The maximum safe rate of SO4-S that can be applied with the seed for cereal crops with a 10 per cent seed-bed utilization (SBU) is 25 to 30 kg/ha (23 to 27 lb/ac) of SO4-S, assuming phosphate fertilizer is also seed-placed, and depending on soil moisture conditions and the opener used.
For oilseed crops, the seed-placed SO4-S rate should not exceed 10 kg/ha (9 lb/ac) when a seeding implement that places the seed and fertilizer in a narrow band (10 per cent SBU) is used, assuming phosphate fertilizer is also seed placed.
For peas, seed-placed P205 rates should not exceed 20 to 25 kg/ha (18 to 23 lb/ac), especially when using a seeding implement that places the seed and fertilizer in a narrow band (10 per cent SBU).
Single and double disc no-till drills may cut a fairly narrow furrow and place the seed and fertilizer in close proximity in the bottom of the furrow. These drills place the fertilizer in a concentrated band close to the seed and are most likely to result in damage to sensitive crops.
Hoe-type openers cut a wider furrow, and the seed and fertilizer are somewhat more scattered across the bottom of the furrow. This scattering in a wider band results in a lower concentration of fertilizer close to the seed and is less likely to cause damage to sensitive crops such as canola or flax when high rates of fertilizer are applied.
Seeding implements that scatter the seed in wide bands (air seeders with sweeps) reduce the seed-fertilizer contact. As a result, higher seed-placed (10 per cent SBU) rates can be used safely. However, because of the wide variation in furrow opener design and spacing, as well as the width of the seed row, specific maximum safe seed-placed (10 per cent SBU) rates have not been determined for all opener types. Generally, rates can be increased by 25 to 50 per cent over the safe rates for a 10 per cent SBU.
Summary
Sulphur is an essential plant nutrient that is of particular importance to crop production in Alberta. Sulphur deficiency is common, particularly in central and northern Alberta soils, and is increasing.
Sulphur deficiency symptoms vary between crops. Symptoms of S deficiency in canola include lighter green colour of newer leaves on most crops. With canola, leaf cupping, purpling and underdeveloped pods are indications of more severe deficiency. If applied early enough, a broadcast application of ammonium sulphate can correct S deficiency.
Fertilizer recommendations should be based on representative soil samples taken in increments of 0 to 6, 6 to 12 and 12 to 24 inches. Fertilizers that supply S in the sulphate form are immediately available to crops. Elemental S� fertilizers must be surface applied where they can disperse before incorporation and be oxidized to plant-available SO4-S. Depending on soil type and environmental conditions, this conversion will take place from within one year to more than four years.
Prepared by
Ross H. McKenzie Ph.D., P. Ag
Sr. Research Scientist - Agronomy
Alberta Agriculture and Rural Development
Previous Agdex prepared by
Mike Hall and Elston Solberg
Photo credits
Figure 2b - Phil Thomas
Figure 4a and 5a - Agronomy Unit collection
All remaining photos taken by Mike Hall
More information, contact
Alberta Ag-Info Centre
Call toll free 310-FARM (3276)
Source: Agdex 542-10. Revised February 2013. |
|