| |

Samplers | Choosing a Sampling Location Checklist | Sample Bottles | Sample Preservation | Field Sampling Data Sheets
Samplers
Dugouts and Ponds
Samples from dugouts or ponds may be taken directly from the source or from a faucet or valve supplied by the water body. Although it is more convenient to sample from a faucet or valve this is not always desirable. Depending on the goal of the analysis it may be better to sample directly from the source even when a faucet is available.

The following devices can be used to obtain water samples directly from the dugout or pond.
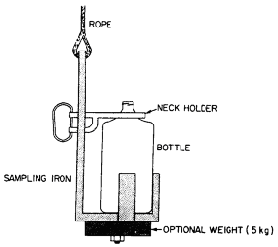
Sampling Iron
A sampling iron is a device made of iron or steel and painted with a rust inhibitor. Typically, it uses a 2-L sample bottle, but smaller bottles may be used. The sample bottles are placed in the sampler and secured by a neck holder. A sample is obtained over the entire depth (depth-integrated sample) by allowing the sampler to sink to the desired depth at a constant rate and then retrieving it at approximately the same rate. The rate should not allow the bottle to fill completely until it reaches the surface.
These types of samplers are relatively easy to make in the farm shop. Usually welding a few pieces of steel will provide an adequate sampler. Other non-leaching materials and variations of the sampler design other than that described here can also be effective.
The sampling iron is most useful near the centre of the dugout or pond when sampled from the side of a boat or off of a dock or other support structure. The necessary safety precautions need to be taken when using this sampler or performing any other type of work on or near open water.
Sampling Tube
A sampling tube is usually constructed from some form of potable water tubing (i.e. Tygon) fitted to a one-way foot valve. As the tube is lowered into the water, the foot valve opens, allowing water to enter the tube. The tube is lowered at a consistent rate to the desired depth. Once the tube is pulled upwards to retrieve the sample, the foot valve closes, keeping the water in the tube.
The sampling tube is most useful near the centre of the dugout or pond when sampled from the side of a boat or off of a dock or other support structure. The necessary safety precautions need to be taken when using this sampler or performing any other type of work on or near open water.
Sampling Stick
In contrast to the sampling iron and sampling tube, a sampling stick is easily used from the shoreline of a dugout or pond. The length of the stick gives the extra reach to obtain a representative sample from the middle of the water body. This type of sampler can be purchased but can be easily constructed using a long shaft with some form of sample container attached to its end. Depending on the length of the shaft, a 1 to 2 litre sample volume should be sufficient for most applications. Too large of a sample container will result in a very heavy and awkward sample to handle. The shaft may be made to be of a fixed length or extendable. The sample container may be fixed-solid or simply tied to the end of the sampling stick.
The sampling stick should be used to obtain a sample from the approximate upper two thirds of the dugout or pond. The sample should not simply be taken directly from the surface as this water will not be as representative of a sample during most conditions unless a sample a surface growth is required, or if water is being used from the surface.
Groundwater Systems
Sampling devices used in groundwater monitoring should consider well diameter and yield, as well as limitations in the lift capacity of the sampling devices and the effect on the analytes in the water sampled from the materials in the sampling devices. Commonly used devices include electric submersible pumps, bailers, suction-lift pumps, and positive displacement bladder pumps. Bailers are often used to both purge and sample small diameter shallow wells.

Table 3: Water-Quality Sampling Devices for Monitoring Wells
| Type | Advantages | Disadvantages |
| Bailer |
- Can be constructed in a wide variety of diameters
- Can be constructed from a variety of materials
- No external power source required
- Very portable
- Low surface-area-to-volume ratio, resulting in a very small amount of outgassing of volatile organics while sample is contained in bailer
- Easy to clean
- Readily available
- Inexpensive
|
- Sampling procedure is time consuming; sometimes impractical to properly evacuate casing before taking samples
- Aeration may result when transferring water to the sample bottle
�
�
�
�
�
� |
|
|
|
|
|
|
|
| Suction-lift Pump |
- Relatively portable
- Readily available
- Inexpensive
|
- Sampling is limited to situations where water levels are within about 20 ft of the ground surface
- Vacuum effect can cause the water to lose some dissolved gas
� |
|
|
| Air-lift Samplers |
- Relatively portable
- Readily available
- Inexpensive
- Suitable for well development
|
- Causes changes in carbon dioxide concentrations; therefore this method is unsuitable for sampling for pH-sensitive parameters
- In general, this method is not an appropriate method for acquisition of water samples for detailed chemical analyses because of degassing effect on the sample
- Oxygenation is impossible to avoid unless elaborate precautions are taken
� |
|
|
|
| Gas- operated Pump |
- Can be used in diameters as small as 1 inch (25.4mm)
- Can be constructed from a variety of materials
- Relatively portable
- Reasonable range of pumping rates
- Driving gas does not contact water sample, eliminating possible contamination of gas stripping
|
- Gas source required (bottled gas and/or compressor)
- Large gas volumes and long cycles are necessary when pumping from deep wells
- Pumping rates are lower than those of suction or jet pumps
- Commercial units are relatively expensive
� |
|
|
|
|
Submer-
sible Pump |
- Wide range of diameters
- Constructed from various materials
- 12-volt pump is highly portable; other units are fairly portable
- Depending on size of pump and pumping depths, relatively large pumping rates are possible for wells larger than 2 inches (51 mm) in diameter
- Readily available
|
- With one exception, submersible pumps (see points below) are too large to fit in 2- inch (51 mm) diameter well casings
- Conventional units are unable to pump sediment-laden water without incurring damage to the pump
- 1.75 inch (44.5 mm) pump delivers low pumping rates at high heads
- Smallest diameter pump is relatively expensive
|
Edited from: Driscoll, F.G. 1995. Groundwater and Wells, Second Edition. Johnson Screens, St. Paul, Minnesota.
Choosing a Sampling Location Checklist
| Sampling Guidelines | Reason |
- Avoid faucets that are seldom used
|
|
- Avoid sampling from a dead-end main
|
|
- Avoid a faucet that leaks around the stem
|
- The faucet may become contaminated
|
- Avoid any faucet that is dusty, dirty or corroded
|
- Possible sources of contamination
|
|
|
- More susceptible to contamination in the swing connection
|
- Avoid faucets that cannot deliver a smooth stream of water
|
|
- Avoid sampling from a flexible hose, garden hose
|
|
- Avoid sampling from faucets with aerators or screen
|
|
- �Avoid sampling from faucets that have a point-of-use carbon filter attached
|
|
Avoid sampling from:
- a dripping faucet
- a frost free yard hydrant or wall hydrant
- an outside hose bib with an unremovable vacuum breaker
- a faucet with an unremovable aerator
- a metal fixture with external plastic or rubber inserts
|
|
|
|
|
|
|
- Choose cold water faucets only
|
- Bacteria may be affected, if hot water is tested
- The chemistry of the water may be changed
|
- Choose a faucet that is NOT connected to the building’s water softener
|
|
- Choose a smooth-end faucet over a threaded-end faucet
|
- Possibility of contamination around the threads
|
Sample Bottles
The specific sample bottles required for the type of testing you request will be provided to you by the laboratory. They are usually well labelled as to the type of sample they are to contain as well as any special instructions necessary. Ask the laboratory staff if you have any questions regarding filling the sample bottles and the preservation techniques required for the different types of bottles. You can also ask the laboratory if it is possible to use your own appropriate sample bottle that you may have available at the time depending on the tests that you require.

An example of the type of sample preservation techniques include:
General chemistry bottles - no preservative, stored at a cool temperature (i.e. 4�C)
Metals - usually use a nitric acid solution as a preservative
Nutrients - usually use a sulphuric acid solution as a preservative
These are only examples. You must check with the specific bottles, preservation techniques, sampling and shipping instructions that the laboratory has provided to you.
Routine care should be taken to prevent bottle and other glassware breakage during sampling; for example, over-tightening of tops, and improper packaging and storage of glass sampling bottles may result in breakage during transport.
Sample Preservation
Unique Problems Associated with Water Samples
Water samples are in a chemically dynamic state, and the moment they are removed from the sample site, chemical, biological, and/or physical processes that change their compositions may begin. Analyte concentrations may become altered due to volatilization, sorption, diffusion, precipitation, hydrolysis, oxidation, and photochemical and microbiological effects.
Free chlorine in a sample can react with organic compounds to form chlorinated by-products. Some treated drinking water and treated wastewater is likely to contain free chlorine.
Samples with photosensitive analytes, such as polynuclear aromatic hydrocarbons, chlorophenols, and bromo- or iodo-compounds, should be collected and stored in amber glass containers to protect them from light.
The composition of water samples may also change because of microbiological activity. This is especially prevalent with organic analytes in wastewater subjected to biological degradation. These samples (and samples containing organic analytes in general) should be cooled immediately, stored, and shipped at a low temperature (about 4�C).
Inorganic analytes may also form salts that precipitate. These precipitates may adhere to the sides of the collection bottle if the sample is not prepared properly and will prevent proper analysis. The most common occurrence is precipitation of metal oxides and hydroxides due to metal ions reacting with oxygen. This precipitation is prevented by adding nitric acid; the combination of a low pH (less than 2) and nitrate ions keeps most metal ions in solution.
Waters with cyanides or sulphides require a sodium hydroxide preservative to be added to ensure that hydrogen cyanide or hydrogen sulphide gas is not evolved. Waters with ammonia are preserved by adding sulphuric acid.
Sample preservation is required to maintain the integrity of the sample. This simply means the water being tested in the laboratory should be as representative as possible of the water that was sampled.
Sample Preservation Techniques
Careful collection and handling of samples must be made to minimize errors. The objective is to provide the laboratory with a set of samples that closely represent the aquatic environment from which they are taken. To ensure consistency and efficiency, sample handling, preservation, storage, and transportation procedures must be properly and accurately documented and adhered to.
The stability of analytes depends on how well the samples are preserved. Preservation instructions must specify proper containers, pH, protection from light, absence of headspace, chemical addition, and temperature control. The chemistry of all analytes must be considered, recognizing that certain reactions may still occur under recommended preservation conditions.
Holding time is the length of time a sample can be stored after collection and preservation, and before preparation and analysis, without significantly affecting the analytical results. Holding times vary with the analyte, preservation technique, and analytical methodology used. Maximum holding times are specified by the method used, and must be taken into account when sampling and analysis protocols are being developed.
Samples preserved by cooling should be refrigerated or sealed and placed in wet ice; blue ice, a synthetic glycol packaged in plastic bags and frozen, is acceptable for maintaining low temperatures. These placed in a common cooler, usually supplied by the laboratory is normally sufficient.
Preservation methods are limited generally to pH control, chemical addition, refrigeration, and sometimes freezing.
Always read the sampling instructions provided by the laboratory and ask them for additional information if unsure.
Safety Precautions when Handling Chemical Preservatives
Reviewing the appropriate Material Safety Data Sheets (MSDS) usually provided or obtainable by the laboratory, provide safety information required for handling chemicals.
Acids and bases used to preserve water samples should be stored and handled with care. Care must be exercised to avoid inhaling vapours or direct contact with skin, eyes, and clothing. Gloves and sometimes an apron should be used when working with these chemical preservatives.
Skin which has been in contact with acids or bases should be washed immediately with plenty of water. After the skin has been washed, the contaminated area may be swabbed with a neutralizing solution. This procedure should be followed by a second washing with soap.
If any chemicals enter the eyes, they should be rinsed immediately with plenty of water. Rinse the outside of the eyes as well. It may be necessary to hold the eyelids open during the wash procedure. Continue rinsing for several minutes. After first aid, all eye injuries must be treated professionally. Consult medical services if deemed necessary and/or contact the laboratory for additional information.
Field Sampling Data Sheets
It can be helpful to use field sampling sheets to record particular details of the sampling, particularly if you are obtaining a large number of samples. You can determine the type of information to record that would best benefit your operation. Record keeping of the sample ID number, location, date, time, type of sample, and method of preservation are some basic things you will want to keep track of. You may also want to record the name of the person performing the sampling, the weather, flow rates, flow meter readings, general and particular observations, as well as any other information which may be useful to you.
It is a good idea to keep this information in a centralized place such as a file folder, log book, or binder as well as a computer file. It can be useful to compare the field sampling information with the water quality results when you receive the analysis back from the laboratory. |
|