| | Introduction | What is biological control? | How are biological control agents identified and introduced? | Advantages and limitations? | A prairie success story | Biological control work in progress | How to obtain biological control agents
Introduction
There are some very tough weeds on the prairies. They compete with valuable crop and forage plants and threaten many native plant species.
Many of these weeds have been very expensive and difficult, if not impossible, to control with more traditional methods. In some instances, the chemicals used for control are non-selective compounds, which will also damage non-target plants and may leach out of sandy or gravelly soils, or compounds that give top growth control only. In addition, because of leaching, chemicals cannot be used on weeds that grow close to bodies of water.
Many problem weeds are abundant in rangeland and along riverbanks and gullies, so the use of non-chemical control methods such as mowing or cultivation can be limited by the topography and size of these areas. These difficulties have prompted scientists to investigate a third alternative: the biological control of weeds.
What is Biological Control?
Biological control covers two key concepts: the deliberate use of a weed's "natural enemies" to suppress its population and the use of these live organisms to maintain this lower population density. A weed's natural enemies may be arthropods (insects, mites and their relatives), bacteria or fungi. These "control agents" feed upon or cause disease in the weed, thereby limiting its growth, reproduction and spread.
There are two distinct primary approaches to weed biocontrol: classical and inundative.
Classical (inoculative) biocontrol involves the release of a relatively small number of control agents. These agents feed on the weed, reproduce and gradually suppress the weed as their population grows. For this approach, arthropods are generally used as control agents.
Inundative biocontrol. In this type of biological control, large quantities of a control agent, generally a pathogen (a bacteria or fungus that causes disease in a weed) are applied to weeds in much the same manner as a chemical herbicide would be.
How are Biological Control Agents Identified and Introduced?
Step 1: Identifying target weeds
To be considered a good candidate for biological control, a weed should be:
| 1. | non-native |
| 2. | present in numbers and densities greater than in its native range and numerous enough to cause environmental or economic damage. The weed should also be present over a broad geographic range |
| 3. | have few or no redeeming or beneficial qualities |
| 4. | have taxonomic characteristics sufficiently distinct from those of economically important and native plant species
for classical biocontrol, the weed should occur in relatively undisturbed areas to allow for the establishment of biological control agents. |
| 5. | Cultivation, mowing and other disturbances can have a destructive effect on many arthropod biocontrol agents. Inundative biocontrol agents such as bacteria and fungi are less sensitive to these types of disturbances so may be used in cropland |
Step 2: Identifying control agents and assessing level of specialization
Scientists observe weeds in their areas of origin (Asia and Europe in the case of many weeds in Canada) and collect the insects and other organisms attacking the plants and affecting their survival. Most of the pathogens being examined as biocontrol agents are native to North America.
These organisms are subjected to a multi-level screening process to assess their host range and their effect on the weed. These screening efforts do two things: they ensure the safety of any valuable crop, forage or native plant species that the agents may encounter when released, and they assess the efficacy of the agent.
Assessing the host range (how specific the potential biocontrol agent is to a particular plant) is probably the most important step in this process. Over very long periods, some plants and herbivores have evolved to form very close associations. Plants have developed a number of defenses, such as toxic chemicals, that plant-eating organisms (herbivores) must overcome. Some herbivores have evolved the ability to bypass only certain host plant's defenses such that they cannot feed or develop on anything else.
To find out how specialized a particular agent is, scientists collect and expose them to a wide assortment of plants. These plants include crop and forage species as well as species native to the intended release area, especially if the species are close relatives of the weed. Screening potential biological control agents ensures that only those with a very narrow host range (i.e. those that represent no threat to crop, forage or native species) are released.
Step 3: Controlled release
All biological control agents must be approved under the Plant Protection Act and are reviewed by an advisory panel of Agriculture and Agri-Food Canada before they can be released. Following federal approval, the agents are released on their target weeds at selected experimental sites, which are closely monitored. Data from these sites help to assess both the agents' potential for survival under field conditions in western Canada and their potential to cause damage to the target weed.
Step 4: Full release and identifying optimal release sites
If the agents survive our harsh climate and damage or suppress the weeds in the controlled tests, they can be released on other weed infestations.
The agents are released on as many varied sites as agent numbers, time and resources will permit. Note that instructions for the proper method and timing of agent releases should be followed closely, and these instructions will vary with the species of agent. The agents are then carefully monitored. Release on a large variety of sites allows scientists to find the habitats best suited to each species.
Many types of biological control agents spend at least part of their lives underground and can be very sensitive to the soil types and conditions present in the various areas. For example, the black dot leafy spurge beetle, Aphthona nigriscutis, spends its entire larval stage underground feeding on leafy spurge roots. Information gathered from a number of release sites indicates that these beetles prefer dry, sandy soils and will not do nearly as well in soils with higher moisture levels.
Step 5: Monitoring release sites (classical)
Release sites should be monitored periodically to assess the size of the biological control agent population and the effect of the agent on the weed. A sweep net is particularly useful for sampling insect biological control agents that feed on the foliage of the weed. (Figure 1).

Figure 1. A sweep net helps in monitoring.
Monitoring release sites is very important to determine if agents have established on a site. Some insect biocontrol agents may need two to five years before their populations increase to a sufficient size to have a visible impact on weed numbers.
When the classical control agent is establishing on a weed infestation and increasing its numbers, the site should not be mowed or disturbed. Herbicide may be used along the boundaries of the weed infestation to help contain the weed while the biological agent is increasing in population and spread.
Step 6: Redistribution (classical)
If a biological control agent is released on a site with favourable conditions, its population can grow quickly.
A large number of insects can suppress the weed in this area and may allow for the establishment of an "insectary." This is an area where the insect population has grown to the point where the insects can be collected in very large numbers. Insects collected from these sites can be distributed to other weed-infested areas. Once established, the insects can suppress the weeds in these areas as well. An important point to remember is that it can take years before a biocontrol agent's population reaches this size. The agents should not be collected for redistribution before this time.
Step 7: Maintaining control agent populations (classical)
What happens to the control agents as their food supply dwindles over time? Because the weeds are never completely eradicated, a small population of insects stays on the few remaining plants. Insects that cannot be supported by the weed population at this lower level either move to new stands or die.
If weed numbers increase for some reason, the number of insects will increase as well. Weed numbers fluctuate around a new, much lower equilibrium point after control is implemented (see Figure 2).
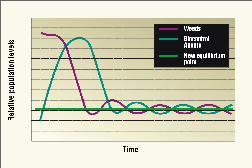
Figure 2. Relative weed and biological control agent population size.
Advantages and Limitations?
Biological control is advantageous because of its selectivity; there is little danger of damage to non-target plant species. Biological control agents are also very effective in inaccessible areas.
Another attractive feature of biological control is its negligible environmental impact. This weed control method does not bring any of the problems associated with herbicide residues, contaminated groundwater and weed resistance to herbicides.
Individual applications of classical biological control are also potentially much less expensive over time. A small number of biocontrol agents can, once established, grow to very high densities and provide continuous control of a weed over a large area.
When the cost of development is considered, classical biocontrol is generally less expensive than chemical control.
Classical biological control does have its limitations, however. It lacks the immediacy of chemical control. Populations require time to become established, so signs of weed suppression are rarely evident in the first year. Screening work (determining the selectivity and effectiveness of a biocontrol agent) is also very time consuming and is subject to limited funding.
A Prairie Success Story
Herbivorous insects have shown the greatest potential as classical biological control agents, and most of the work on the biological control of weeds on the prairies involves their use (Figure 3).
 |  |
| 1990 | 1992 |
| Figure 3. Leafy spurge biocontrol (Edmonton). |
Evaluating the effectiveness of these insects to control some of the worst weeds on the prairies is the focus of much research by Alberta Agriculture, Food and Rural Development, Saskatchewan Agriculture and Food, Manitoba Agriculture, the Alberta Research Council and Agriculture and Agri-Food Canada, in cooperation with agencies from around the world.
The most successful insect to be used in the prairies is the black dot spurge beetle (Aphthona nigriscutis). Release of these insects on leafy spurge has resulted in a 99 per cent reduction in spurge stand density in one area and a corresponding 30-fold increase in grass biomass after four years.
Leafy Spurge (Euphorbia esula) is a deep-rooted, noxious perennial weed that was accidentally introduced to North America in the early 1800's (Figure 4). The plants grow to a height of 1 metre, have long, thin, dark green leaves and can be identified from a distance by their distinctive yellow-green flowers. Since its introduction, it has spread to become a very serious problem on rangeland, pasture and grassland throughout the southern prairies and is gradually working its way north.

Figure 4. Leafy spurge has become a serious problem.
Leafy spurge is very competitive and easily out-competes many forage and native plant species. The juice of the plant is a white, milky latex that may cause mouth and throat blistering in cattle and contact dermatitis in people. Ingestion of large amounts of leafy spurge has been suspected of causing death in cattle.
Cattle avoid spurge-infested areas, greatly reducing the livestock carrying capacity of infested range and pastureland. Leafy spurge has proven to be very difficult and expensive to control with herbicides and virtually impossible to control with cultural techniques.
Originating in Europe, the black dot spurge beetle (Aphthona nigriscutis) measures 2 to 3 mm in length. Adult beetles feed on the foliage of the weed and larvae feed on the roots (Figure 5). The larvae cause most of the damage to the plant, both directly through their root and root hair feeding, and secondarily by creating feeding wounds that provide an entry for various disease-causing organisms. The black-dot spurge beetle produces one generation per year. Each adult female is capable of producing about 150 offspring in a growing season. This control agent does best in dry, sandy soils (Figure 6).

adult | 
larva |
Figure 5. Black dot spurge beetle.

Figure 6. Good black dot spurge beetle release site.
Several other spurge beetles have shown promise in controlling this noxious weed: Aphthona lacertosa and Aphthona czwalinae, the brown-legged and black spurge beetles (Figure 7) and Aphthona cyparissiae, the brown dot spurge beetle.

Figure 7. Brown-legged spurge beetle.
The brown-legged and black spurge beetles have been available in numbers for only a short time, so characterization of their habitat requirements is ongoing. Initially, they seem to prefer relatively moister and loamier sites than A. nigriscutis. The brown dot spurge beetle shows a preference for slightly moist, sandy soils in open sunny areas. Research is continuing to find insects that will attack leafy spurge in forested areas.
Biological Control Work in Progress
Other available biological control agents
 |  |
| Figure 8. Scentless chamomile (Matricaria perforata) with the seed weevil Omphalapion hookeri. |
 |  |
 | 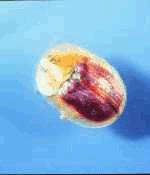 |
| Figure 9. Bladder campion (Silene vulgaris) with the foliage, flower and seed-feeding tortoise beetle Cassida azurea. |
Agents currently being tested:
- scentless chamomile (Matricaria perforata) with the Austrian stem-mining weevil Microplontus edentulus and the gall midge Rhopalomyia sp.
- yellow toadflax (Linaria vulgaris) and Dalmatian toadflax (L. dalmatica) with the stem-mining weevil Mecinus janthinus and yellow toadflax with the root-mining moth Eteobalea serratella (Figure 10)
 |  |
| Figure 10. Yellow toadflax (Linaria vulgaris) with the stem-mining weevil (Mecinus janthinus). |
- field bindweed (Convolvulus arvensis) with the gall-forming mite Aceria malherbae
- cleavers (Galium spurium) with the gall mite Cecidophyes rouhollahi
- hound's-tongue (Cynoglossum officinale) with the root weevil Mogulones cruciger
- purple loosestrife (Lythrum salicaria) with foliage feeding beetles of the genus Galerucella
Tansy (Tanacetum vulgare) is being evaluated as a possible candidate for biological control.
Trials focusing on inundative biological control are also underway. The first pathogen to be registered as a bioherbicide was the fungus Colletotrichum gloeosporioides f.sp malvae (Figure 11) for control of round-leaved mallow (Malva pusilla) in field crops (Figure 12). This bioherbicide was formerly licensed for commercialization under the tradename BioMal. Currently, it is licensed to Encore Technologies LLC with a tradename to be determined for commercial release in the near future.
Other target weeds for inundative biological control are wild oat, green foxtail, Canada thistle, cleavers and scentless chamomile.
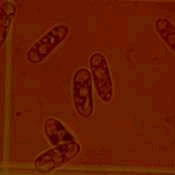 |  |
| Figure 11. Fungal spores (left) of Colletotrichum gloeospotioides f.sp. malvae causes stem lesions and girdling (right) on round-leaved mallow (Malva pusilla). Spores are about 1 to 2 microns (a micron is one millionth of a metre). |

Figure 12. Lentils treated with Colletotrichum gloeosporioides f.sp. malvae (lower left) and the untreated control (lower right) three weeks after application.
How to Obtain Biological Control Agents
Farmers, ranchers and landowners with any questions or an interest in obtaining these biological control agents should contact:
- local provincial government crop specialist or extension agrologist
- municipal agricultural fieldman
- Dr. Alec McClay, Alberta Research Council (Vegreville) (780) 632-8207
- Dan Cole (780) 422-0919, Agronomy Unit, Alberta Agriculture and Food (Edmonton)
- Clark Brenzil (306) 787-4673, Sustainable Production Branch, Saskatchewan Agriculture and Food (Regina)
- Kim Brown (204) 745-5671, Crop Diversification Section, Manitoba Agriculture (Carmen)
- Dr. Rose DeClerck-Floate or Dr. Rob Bourchier, Agriculture and Agri-Foods Canada (Lethbridge) (403) 327-4561
- Dr. Karen Bailey or Dr. Susan Boyetchko, Agriculture and Agri-Food Canada (Saskatoon) (306) 956-7200
Contact any of the above if landowners are interested in hosting experimental sites or have additional questions.
Photos courtesy of Alberta Agriculture and Food; Agriculture and Agri-Food Canada; Alec McClay and Robert B. Hughes, Alberta Research Council.
Source: Agdex 641-1. February 2000. |
|